- Product - Profile
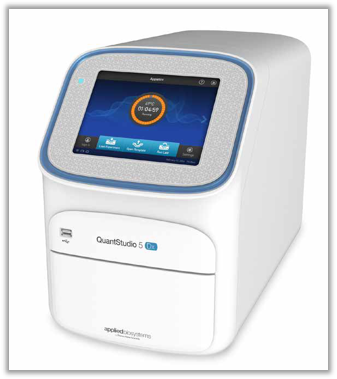
QuantStudio 5 Dx Real-Time PCR System
Accurate, sensitive, and reliable real-time PCR system designed for the clinical laboratory
The Applied Biosystems QuantStudio 5 Dx Real-Time PCR System is designed to streamline workflows and minimize training needs, helping you get clinical answers quickly by integrating seamlessly into your established workflow.
Features
Results you can trust
- Detect differences in target quantity as small as 1.5-fold in singleplex reactions and obtain 10 logarithmic units of linear dynamic range
Designed with the clinic in mind
- An interactive diagnostic instrument with short run times and minimal maintenance that also uses existing plastic consumables for added convenience in clinical laboratories
QuantStudio performance
- High reliability, sensitivity, and accuracy with an intuitive and simple-to-use interface that allows users of any experience level to operate the system with ease
Simple, powerful software
- A single, easy-to-use software interface allows users to set up a run, lay out assays, control the instrument, and conduct plate analysis
IVD Test mode
- The in vitro diagnostic (IVD) test menu allows only authorized IVD tests to be run, reducing the risk of unauthorized use and accidental or intentional misuse
Enhanced security
- Security, auditing, and e-signature functionalities that enable you to comply with regulations, including 21 CFR Part 11
Watch the QuantStudio 5 Dx in Action